Spark R&D Montana Mask Information Page
In response to the COVID-19 pandemic, Spark R&D began producing injection-molded plastic parts for the Montana Mask. Spark R&D, a Bozeman, Montana based manufacturer of splitboard bindings and accessories, was in a unique position to rapidly help create these parts with in-house design, machining, and injection molding capabilities.
As of 2/9/2021
The Montana Mask Story
Background of the 3D Printed Mask
The Montana Mask was designed out of Billings, Montana by Dusty Richardson, MD, Spencer E. Zaugg, DMD, and Colton Zaugg in the wake of PPE shortages due to the COVID-19 crisis. It was designed to be an open-source, grassroots movement, where anyone can download the files and print the masks on a 3D printer. The idea is that anyone with access to a 3D printer could help to supply healthcare workers who were not able to secure conventional PPE. The story aired on CNN on March 23, 2020: https://www.cnn.com/2020/03/23/us/coronavirus-3d-printed-medical-supplies-trnd/index.html
Since that time, the file has been downloaded in 150+ countries and with the flurry of phonecalls and emails, it became abundantly clear that access to PPE worldwide was even more limited than anyone could have imagined.
The move to injection-molded Montana Masks
Will Ritter, owner of Spark R&D, heard of the CNN story and wanted to get involved. While Spark R&D does indeed have a 3D printer and would have been more than willing to join the printing efforts, Will reached out to Dr. Richardson with an idea. With the volume of masks needed and the short window before healthcare workers run out, injection-molding the masks would be a much more efficient and timely solution. 3D printing masks takes around 3 hours for one mask. Injection molding could be as quick as one every 45 seconds. Spark R&D could use the 3D files provided and re-design them for an injection molded mask. With in-house design, mold-making, machining, and injection molding capabilities a prototype was made in just four days and the final product was approved on day six, on March 31, 2020. The first 2400 masks were delivered to the Billings Clinic on April 4th, 2020.
More information about the mask - https://www.makethemasks.com/
More about Spark R&D - https://www.sparkrandd.com/company/
Spark R&D Montana Masks Blog - https://www.sparkrandd.com/the-montana-mask-project
Teton Gravity Research Mask Story - https://bit.ly/3b0JFEB
The Snowboarders' Journal - https://bit.ly/2VfqjpU
Billings Gazette Montana Mask Story - https://bit.ly/39DTbMm
Bozeman Daily Chronicle Story - https://bit.ly/39S5N2H
Mask Related Photos - https://bit.ly/2UwRBaK
Real Talk Snowboarding Podcast - https://bit.ly/2YUTr8e
Media Inquiries:
- Dan Ventura, Marketing Manager
- E: Dan@sparkrandd.com

- Filters still available:
- We have pre-cut KN95 filter material available for sale in 10 packs, for $2.50 each.
- We have instructions for using and reusing surgical or N95 masks and precut filtration material for use with Montana Masks. Follow the links or click on the graphics below. Reuse instructions as recommended by the CDC.
- Video tutorial for modifying a surgical mask - https://youtu.be/EK2k8oBbRVY
Montana Mask Specs:
- Important Note: The Montana Mask is not an FDA or NIOSH approved N95 replacement. These masks are intended for use past crisis capacity if there is no conventional PPE available. Please closely read the info available in this document and on makethemasks.com to decide if they are right for you.
***Please read the legal disclaimer below
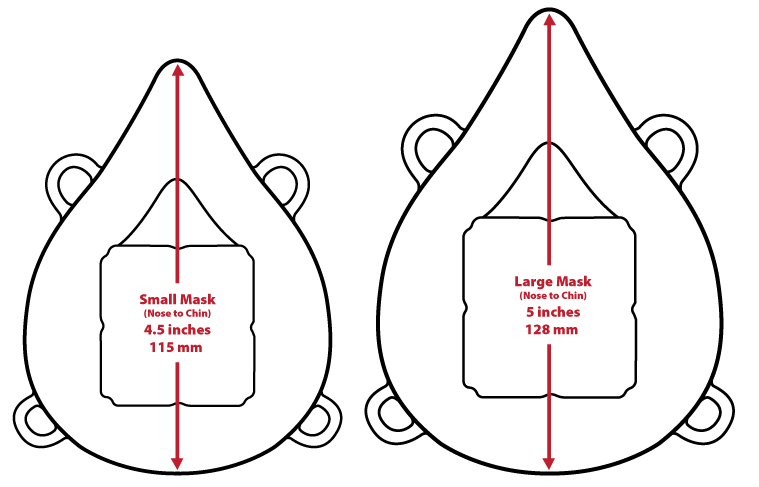
- Sizes:
- Small Mask - Nose to chin measures 4.5 in / 115mm
- Large Mask - Nose to chin measures 5 in / 128mm
- Click to enlarge
- Mask materials: Spark R&D made masks out of an FDA compliant TPE material. Other shops may use other materials.
- Sterilization: These masks can be put into an autoclave or otherwise wiped clean for sterilization.
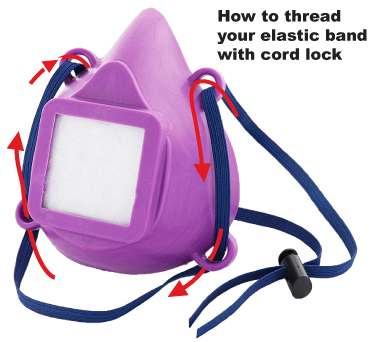
- Straps: Masks do not come with straps. You can do it yourself with a 36" piece of elastic and a cord lock.
click to enlarge
For Manufacturers:
We are so grateful to have had the support of designers and manufacturers all over Montana and beyond. This project has been a collaborative and open-source effort and the list of thank-yous is long.